Functional and Cognitive Recovery of Patients with Traumatic Brain Injury
Prediction Tree Model Versus General Model
- HyunSoo Oh, RN, PhD and
- WhaSook Seo, RN, PhD
+Author Affiliations
<>1.Corresponding author: WhaSook Seo, RN, PhD, Department of Nursing, College of Medicine, Inha University, YongHyun Dong 253, Incheon, 402-751, Republic of Korea (e-mail: [email protected]).- This article has been designated for CE credit. A closed-book, multiple-choice examination follows this article, which tests your knowledge of the following objectives:
- Recognize the difference between functional and cognitive recovery in the patient with traumatic brain injury (TBI)
- Discuss the role of predictive models in the care of the patient with TBI
- Identify patient variables that can influence cognitive and functional recovery at 1 and 6 months in the patient with TBI
A prediction model for recovery or mortality after brain injury must be quick and simple enough to use without special training.
Traumatic brain injury (TBI) is a leading cause of death and disability.1,2 As a result of TBI, 1.5 million persons die every year3 and nearly 2% of the world population has a long-term or lifelong need for help in performing activities of daily living.4 A patient’s condition during the first few weeks after a TBI is extremely unstable and life-threatening. During such critical periods, accurate neurological assessment is essential for predicting recovery. Many researchers have evaluated various clinical variables as prognostic indicators.5–10
For critical care nurses, data from neurological assessments are particularly important, both for evaluating the severity of TBIs and for planning the short-term management and early rehabilitation of patients with TBI. The care team and the families of patients with TBI are concerned about the prognosis for death/survival and degrees of disability and about possible complications during critical periods. A valid and reliable prediction model can increase understanding of patients’ status and rehabilitation.
Background
Most studies6–8,10–20 of TBI have been conducted to identify significant predictors of mortality or recovery (Table 1⇓). Results of those studies were used to propose clinical variables that influence the recovery of patients with acquired brain injury. On the basis of study results, various prediction models have been proposed (Table 1⇓). However, prediction models tend to include many predictors and therefore become complicated, which limits their clinical utility.
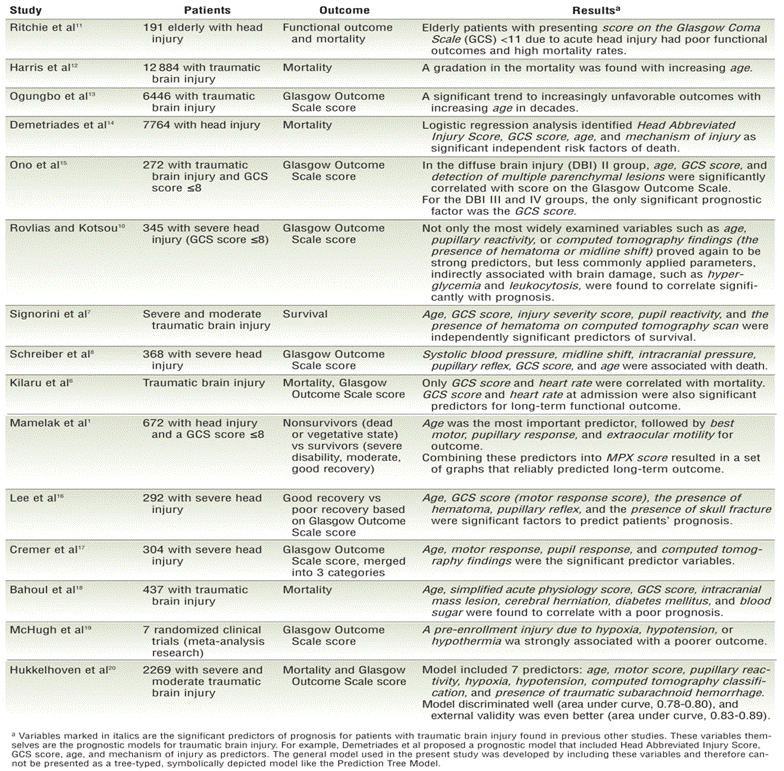
Table 1
Studies on prognostic prediction for patients with traumatic brain injury
Much time or special training is sometimes required to implement a prediction model, depending on the variables included in the model. For example, implementation of a prediction model that includes injury severity as a predictor and requires assessment of injury severity by using the Acute Physiology, Age, and Chronic Health Evaluation II or the Head Abbreviated Injury Score is quite time-consuming. On the other hand, implementation of a model that includes the presence of intracranial hematoma, hematoma size, or the degree of midline shift as a predictor may require special training in interpreting computed tomography scans.
Therefore, to be practical, a prediction model must be simple enough to use without special training or an excessive time commitment.21 Some compromise between simplicity and accuracy must be reached in order for such techniques to be generally applicable.
From this point of view, the Prediction Tree Model (PTM) developed by Choi and colleagues22 has some advantages. This model is fairly concise and includes 4 major indicators (age, pupillary reflex, motor response, and mass lesion) that are routinely or easily accessible (Figure 1⇓). The tree structure shows that the assessment sequence of these indicators varies among patients, and therefore, the PTM can be applied flexibly to different patients.
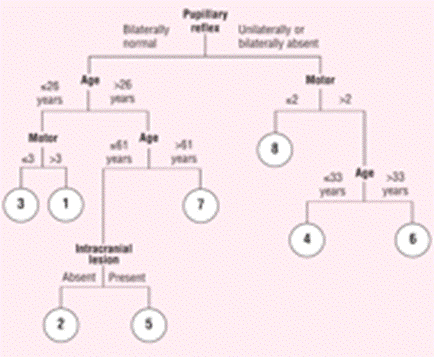 Prediction Tree Model.22 This model includes 4 prognostic indicators (pupillary reflex, age, motor response, and intracranial lesion) and shows the critical point of each indicator. The critical point designates the threshold for the prognostic variable, the critical level beyond which the risk of a poor outcome is substantially increased or decreased. Patients can be split into smaller and smaller subgroups based on the critical levels of prognostic indicators. According to the prognostic grades, there can be 8 subgroups that inversely reflect prognostic levels.
Prediction Tree Model.22 This model includes 4 prognostic indicators (pupillary reflex, age, motor response, and intracranial lesion) and shows the critical point of each indicator. The critical point designates the threshold for the prognostic variable, the critical level beyond which the risk of a poor outcome is substantially increased or decreased. Patients can be split into smaller and smaller subgroups based on the critical levels of prognostic indicators. According to the prognostic grades, there can be 8 subgroups that inversely reflect prognostic levels.
Another strength of this tree model is that it provides information about the critical point of each indicator. The critical point designates a threshold or critical level of the prognostic variable beyond which the risk of a poor outcome is substantially increased or decreased.
Patients with bilaterally normal pupillary reflexes tend to have a good outcome, unless they are more than 26 years old (Figure 1⇑). This result indicates that an age of 26 years is the threshold or critical level for the age factor. The PTM relies on recursive splitting (a statistical classification method with a series of binary splits according to the critical point of each indicator) of patients into smaller subgroups based on the critical levels of prognostic indicators; the patients are first split based on their pupillary reflex, and then subsequently split based on age and motor response. Finally, subjects can be split into 8 subgroups according to their prognostic grades (1–8), which inversely reflect prognostic levels.
Choi and colleagues22 reported that the PTM was statistically significant for predicting the survival rate and recovery status of patients with TBI. However, because Choi et al used a single-item scale, the Glasgow Outcome Scale (5 levels: dead, persistent vegetative state, severely disabled, moderately disabled, and good recovery) to evaluate the degree of recovery, their results can be used only to infer a general status of recovery, not specific aspects of recovery.
Specifically, the value of the PTM for predicting functional and cognitive recovery must be evaluated. Functional recovery is an ongoing process to improve physical performance of activities of daily living, whereas cognitive recovery refers to a mental process to improve memory, attention, problem solving, and judgment. Functional and cognitive recovery can be important outcomes when planning the management and rehabilitation of patients with acute brain injury. Many survivors remain disabled and are discharged to their homes to lead somewhat dependent lives. The value of the PTM for predicting the multiple dimensions of recovery has not been examined.
Aims: The purpose of the present study was to assess the value of the PTM in predicting functional and cognitive recovery. Specifically, this study was conducted to compare the PTM with the general model to verify the value of the PTM in predicting functional and cognitive recovery 1 month and 6 months after admission to the intensive care unit. We developed the general model on the basis of the results of previous other studies (listed in Table 1⇑, studies retrieved by searching CINAHL and PubMed with key words of TBI, prognosis, prediction model, mortality, and recovery). The general model included many significant predictors of outcome after brain injury, which were verified from other studies (marked in italics in Table 1⇑): age, Glasgow Coma Scale (GCS) score, pupillary reflex, the presence of subarachnoid hemorrhage and midline shift, systolic blood pressure, and heart rate at admission. The specific study questions were (1) Is the PTM better than the general model for predicting functional recovery at 1 month and 6 months? and (2) Is the PTM better than the general model for predicting cognitive recovery at 1 month and 6 months?
Methods
Study Design and Data Collection
The participants in this prospective study were 83 patients with TBI admitted to a neurosurgical intensive care unit at a university hospital located in Incheon, South Korea, who had survived at least 6 months after brain injury. Patients with TBI who were at least 18 years old, had impaired consciousness, and had been admitted to the neurosurgical intensive care unit immediately after emergency care were included in the study. We excluded patients with previous physical or cognitive disabilities.
Data related to the previously identified prognostic factors of brain injury (ie, age, GCS score, pupillary reflex, systolic blood pressure and heart rate at admission, diagnosis, computed tomography findings, type of surgery, and relevant medical history such as history of hypertension or diabetes mellitus) were collected by reviewing medical records from September 2002 to December 2004. The outcomes, namely, functional disability and cognitive ability, were evaluated 1 month and 6 months after admission to the ICU. All data were collected by the first author.
Data were collected with permission from the hospital’s institutional review board. Informed consent was obtained from patients or from family members of patients who were unable to give informed consent. We were not always able to obtain informed consent. Data were included only for those patients who had given informed consent. Informed consent was also obtained for the data on 1-month and 6-month recovery. When obtaining informed consent, we provided patients and their families with details about the purpose and process of the present study and explained that all data were confidential.
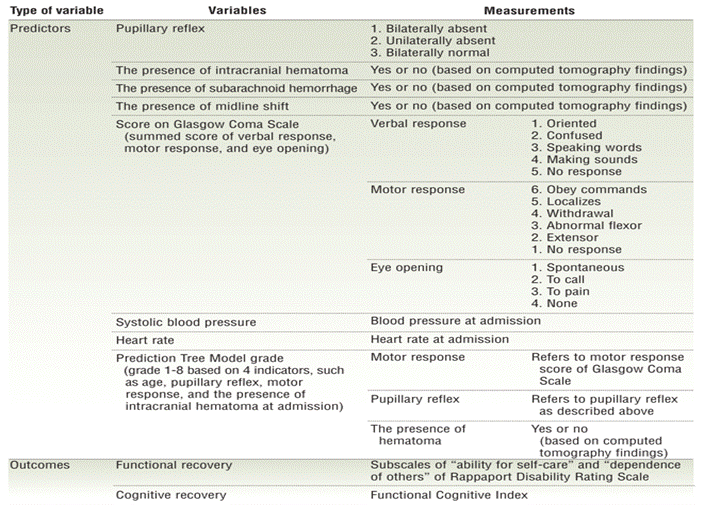
Measurements
The level of consciousness on admission was measured by using the GCS. Pupillary reflex was evaluated by using a 3-point rating scale that had been commonly used in other studies, namely, 1 = bilaterally absent, 2 = unilaterally absent, and 3 = bilaterally normal.5,7,10 The presence of intracranial hematoma, subarachnoid hemorrhage, and midline shift was determined from computed tomography findings. The prognostic grade was measured by using the PTM and classified into 8 grades (1–8) on the basis of the 4 indicators of the PTM: age, pupillary reflex, motor response, and the presence of intracranial hematoma at admission.
As outcome variables, functional disability was measured by using the Rappaport Disability Rating Scale,23 which was originally developed to quantify the degree of disability over the course of recovery after severe brain injury. This 8-item rating scale consists of 4 subscales: “arousability and awareness,” “ability for self-care,” “dependence on others,” and “psychosocial adaptability.”23 In the present study, the arousability and awareness and psychosocial adaptability subscales were not used because the focus of the study was on evaluating functional disability. The arousability and awareness sub-scale is a slight modification of the GCS and reflects impairment ratings for eye opening, communication ability, and motor response. The psychosocial adaptability subscale was designed to predict employability (not restricted, selected jobs, sheltered workshop, not employable).
On the other hand, the self-care subscale and the dependence on others subscale were suitable for evaluating the degree of functional disability because the former reflects the degree of disability in feeding, grooming, and toileting and the latter evaluates the level of physical dependence on others. Therefore, these 2 subscales were used in the present study. In terms of the ability for self-care subscale, feeding, grooming, and toileting functions were each rated as “completely independent” (rating of 0), “partially independent” (rating of 1), “minimally independent” (rating of 2), or “completely dependent” (rating of 3). On the other hand, the ability for self-care subscale was rated as “completely independent” (rating of 0), “independent in special environment” (rating of 1), “mildly dependent” (rating of 2), “moderately dependent” (rating of 3), “markedly dependent” (rating of 4), or “totally dependent” (rating of 5). The maximum score on this scale is 14, with higher scores representing higher levels of functional disability.
The Rappaport Disability Rating Scale is a valid and reliable measure.2,23–25 The score on the Rappaport Disability Rating Scale is a significant predictor of long-term disability in patients with moderate and severe TBI and also shows a significant association with neurophysiological measures of brain dysfunction as reflected in brain evoked potential abnormality scores. The Cronbach α of 0.93 found in the present study also supports the high reliability of the Rappaport Disability Rating Scale.
Cognitive ability was measured by using the Functional Cognitive Index, which was designed to assess attention, communication, behavior/safety, behavior/social, problem solving, and memory. The Functional Cognitive Index is a 6-item, 6-point rating scale. Attention, communication, behavior/safety, behavior/social, problem solving, and memory function were each rated as “no response” (rating of 0), “severe disability” (rating of 1), “moderate disability” (rating of 2), “mild disability” (rating of 3), “minor disability” (rating of 4), or “normal response” (rating of 5). The validity of the Functional Cognitive Index was examined by correlating scores from this scale with scores from the Rancho Los Amigos Scale of Cognitive Function, the most common scale for assessment of cognitive function. The correlation coefficients between these 2 scores were 0.79 at admission and 0.77 at discharge, indicating high validity for the Functional Cognitive Index. The intraclass correlation coefficient, a stringent measure of concordance, of the Functional Cognitive Index was also very high (0.88), signifying high reliability.26 The reliability coefficient (Cronbach α) of the Functional Cognitive Index in the present study was 0.98.
Scores on the Rappaport Disability Rating Scale and the Functional Cognitive Index were measured 1 and 6 months after admission directly or by telephone interview for subjects discharged before 6-month assessments. The reason for evaluating recovery status at 6 months after admission was that physical and neural recovery reach a maximum level at around 6 months after trauma, and therefore, 6-month recovery has been commonly assessed in other studies.2
The first author (H.O.) evaluated functional and cognitive ability 1 month and 6 months after admission. At the 1-month evaluation, most data were collected by direct (face-to-face) assessments because most patients were in the hospital during this evaluation. At the 6-month evaluation, however, data were collected mostly by telephone interview because patients were generally discharged before the 6-month assessments. All instruments used to measure variables are listed in Table 2⇓.
View this table:
Table 2
Instruments used to measure each variable
Data Analysis
Statistical analysis was performed by using SPSS (version 12.0). Descriptive analysis was used to analyze general subject characteristics. Multiple regression analysis was conducted to determine the adjusted coefficient of determination (adjusted R2) of the PTM grade for predicting outcomes, the degree of functional disability and cognitive ability. Simple regression analysis for the curve estimation was also conducted to further analyze whether the relationship between a specific predictor and the outcome variable was linear or nonlinear.
Results
Descriptive Analysis
The mean age of the subjects was 50.99 (SD, 15.56) years, and ages ranged from 19 to 85 years; 71 subjects (86%) were male and 12 (14%) were female (Table 3⇓). Hypertensive patients accounted for 11% and diabetes mellitus for 5% of the participants in the study. Of the 83 patients in the study, 12% had a TBI while under the influence of liquor. Regarding diagnosis, 42% of participants had more than 1 diagnosis: 89% had intracranial hemorrhage; 43% had subdural, 32% had epidural, and 29% had traumatic subarachnoid hematoma; 23% had traumatic intracerebral hemorrhage; and 4.8% had intraventricular hemorrhage. The second diagnosis was included in this analysis (Table 3⇓). Cerebral midline shifts were observed in 24% of the patients. The mean degree of these midline shifts was 7.76 (SD, 5.46) mm, and they ranged from 1.7 to 17.0 mm. Of the 36% of participants who had at least 1 surgery after admission, 60% underwent craniotomy (hematoma removal) and 33% underwent decompressive craniectomy.
View this table:
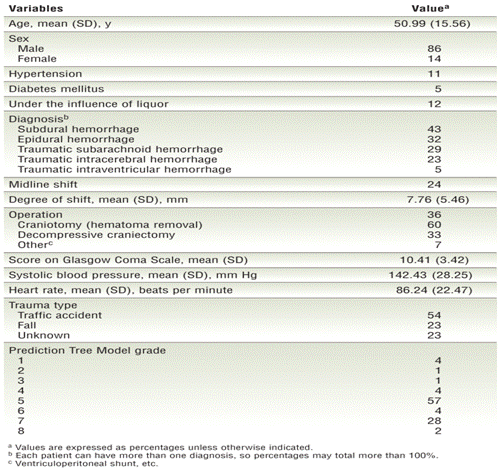
Table 3
Descriptive statistics of the study variables (n = 83)
At admission, mean GCS score was 10.41 (SD, 3.42; range, 3–15), systolic blood pressure was 142.43 (SD, 28.25) mm Hg, and heart rate was 86.24 (SD, 22.47) beats per minute. Patients were injured predominantly during traffic accidents (54%) or falls (23%). Regarding the prognostic grade estimated by using the PTM, participants in the present study were of grade 1 (n=3, 4%), grade 2 (n=1, 1%), grade 3 (n=1, 1.2%), grade 4 (n=3, 4%), grade 5 (n=47, 57%), grade 6 (n=3, 4%), grade 7 (n=23, 28%), and grade 8 (n=2, 2%). Most of the subjects in this study were of grades 5 and 7 (Table 3⇑).
1-Month and 6-Month Functional Disability
The adjusted R2 of the PTM for predicting 1-month functional disability was 0.01, and this result was not statistically significant (F= 1.02, P=.32). On the other hand, the adjusted R2of the general model, which included all related clinical variables, was 0.44, and this result was statistically significant (F =10.46, P<.001; Table 4⇓). The significant predictors of 1-month functional disability were GCS score, pupillary reflex, age, and the presence of subarachnoid hemorrhage (Table 4⇓). Thus poor 1-month functional recovery appears to be associated with a lower GCS score, abnormal pupillary reflex, older age, and the presence of subarachnoid hemorrhage on admission (note the positive and negative signs of β coefficients in Table 4⇓).
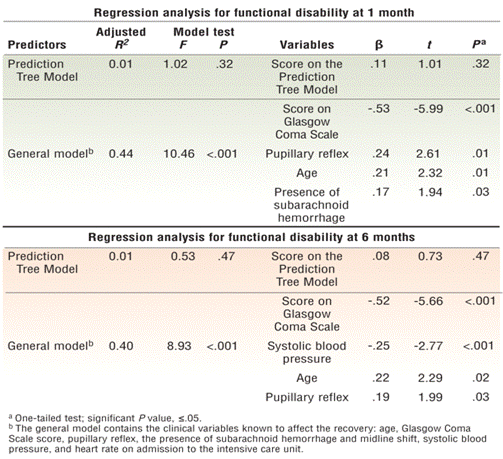
Table 4
Analysis of functional disability at 1 month and 6 months (n=83)
The adjusted R2 of the PTM for predicting 6-month functional disability also was 0.01, which was not statistically significant (F=0.53, P=.47; Table 4⇑). On the other hand, the adjusted R2 for the general model, which included all related clinical variables, was 0.40, which was statistically significant (F=8.93, P<.001; Table 4⇑). The significant predictors of 6-month functional disability were GCS score, systolic blood pressure, age, and pupillary reflex (Table 4⇑). Thus poor 6-month functional recovery appears to be associated with a lower GCS score, a lower systolic blood pressure, older age, and an abnormal pupillary reflex on admission (note the positive and negative signs of β coefficients in Table 4⇑).
Although the results of the present study indicated that a higher systolic blood pressure was positively related to 6-month functional recovery, previous other studies showed that a markedly high blood pressure seemed to have a negative influence on the prognosis. Therefore, to analyze further the relationship between blood pressure and functional recovery, a curve estimation of simple regression analysis was conducted to examine the relationship between systolic blood pressure and 6-month functional disability. Results showed a significant quadratic (U-shaped) relationship between systolic blood pressure and 6-month functional disability (Figure 2⇓). Specifically, 6-month functional disability of patients with a systolic blood pressure on admission of less than 150 mm Hg improved with increases in systolic blood pressure, but the 6-month functional disability of those with a systolic blood pressure on admission of 150 mm Hg or more worsened with increases in systolic blood pressure. In the present study, patients with a systolic blood pressure of 120 to 150 mm Hg on admission showed the best 6-month functional recoveries.
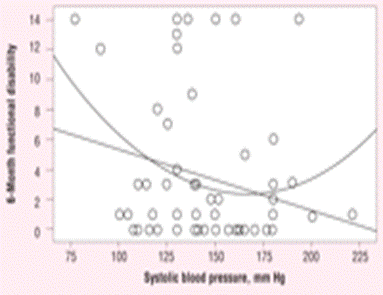
Figure 2
Simple regression analysis of 6-month functional disability (score on Rappaport Disability Rating Scale) with systolic blood pressure for curve estimation (linear: R2 = 0.06, F = 4.94, P = .03; quadratic: R2 = 0.10, F = 4.64, P = .01).
1-Month and 6-Month Cognitive Ability
The adjusted R2 of the PTM for predicting 1-month cognitive ability was 0.02, and this result was not statistically significant (F=0.00, P=.97). By contrast, the adjusted R2 of the general model, which included all related clinical variables, was 0.12, and this result was statistically significant (F=2.02, P=.05). The significant predictors of 1-month cognitive ability were pupillary reflex and heart rate at admission (Table 5⇓). Thus poor 1-month cognitive recovery appears to be associated with abnormal pupillary reflex and a higher heart rate at admission.
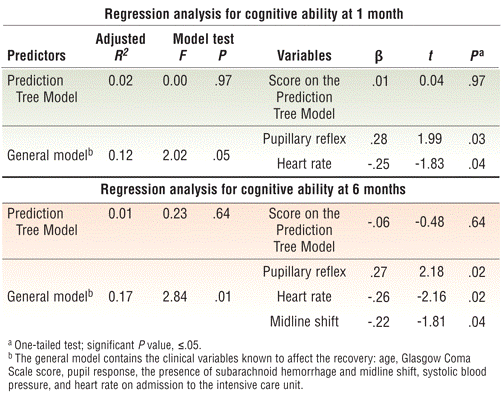
Table 5
Analysis of cognitive ability at 1 month and 6 months (n=83)
The adjusted R2 of the PTM for 6-month cognitive ability was 0.01, and this result was not statistically significant (F=0.23, P=.64). However, the adjusted R2 of the general model, which included all related clinical variables, was 0.17, and this result was statistically significant (F=2.84, P=.01). The significant predictors of 6-month cognitive ability were pupillary reflex, heart rate, and midline shift on admission (Table 5⇑). Thus poor 6-month cognitive recovery appears to be associated with abnormal pupillary reflex, a higher heart rate, and the presence of midline shift on admission.
Discussion
The emphasis on critical care management to decrease mortality and secondary brain injury after TBI has been increasing. The general principle in the management of patients with TBI includes maintaining cerebral oxygenation, decreasing intracranial pressure, and improving neurological function.27 To optimize cerebral oxygenation, the PaO2 should be maintained at 90 to 100 mm Hg and airway support and ventilatory assistance must be provided.28 Patients with elevated intracranial pressure can be treated with hyperventilation, osmotic diuretics, drainage of cerebrovascular fluid, or surgery. Maintaining systolic blood pressure between 100 and 150 mm Hg is important for ensuring that cerebral perfusion pressure is adequate. Barbiturate or anticonvulsant medications also can be used judiciously to decrease cerebral blood flow and metabolism.27,28
The degree of recovery in patients with TBI depends on those acute medical managements. However, many other factors influence recovery. Researchers are continuously pursuing efforts to identify significant predictors of TBI recovery and to develop and verify predictive models. Such efforts have contributed to enhanced recovery of patients with TBI.
Comparisons of the PTM With the General Model
Although the PTM includes the major parameters previously identified as predictors, it did not yield significant results for predicting functional and cognitive recovery at 1 month or 6 months after admission. These poor results may be explained as follows. First, a preponderance of participants were in prognostic grades 5 and 7, which might have affected the performance of the PTM by decreasing variance of the outcome variable (prognostic grade). Multiple regression analysis can be affected by the variance of the variables, that is, the level of dispersion of PTM scores in this study. However, the dispersion of the PTM grade is naturally low (within the range of 1–8). Moreover, the preponderance of grades 5 and 7 in the present study further decreases the variance of the outcome variable.
Second, the PTM was originally developed to predict mortality and general recovery, whereas we used the PTM for predicting functional and cognitive recovery in an effort to determine whether the PTM can be applied to other aspects of recovery. Overall, we found that the PTM has a low validity for predicting functional and cognitive recovery.
Significant Predictors of Functional and Cognitive Recovery
The results obtained by applying the general model showed that good functional recovery appears to be commonly associated with a better GCS score, better pupillary reflex, and a younger age at both the acute (1 month) and post-acute (6 month) stages of TBI. The only difference between the acute and post-acute stages in terms of the factors found to affect functional recovery was that the acute stage was influenced by subarachnoid hemorrhage whereas the postacute stage was affected by the systolic blood pressure at admission; that is, the presence of subarachnoid hemorrhage exerted a negative influence exclusively on 1-month functional recovery, and lower systolic blood pressure at admission exerted a negative influence exclusively on 6-month functional recovery.
GCS score, pupillary reflex, and age, which were frequently reported as significant predictors of mortality, were significant predictors of 1- and 6-month functional recoveries in the present study, and of these, GCS score appears to be the most important predictor (a higher β coefficient value indicates a higher level of predictability regardless of its sign, positive or negative). Some researchers have evaluated motor response by using the motor response subscale score of the GCS rather than the GCS total score, because GCS measurements of verbal and eye opening responses can be inaccurate in practice, particularly in patients who are intubated or have facial edema.8 Regardless, they obtained the same result with both methods.
One of the most well-known predictors of mortality, pupillary reflex, was also a significant predictor of overall functional recovery in the present study. The predictive value of pupillary reflex seems to be fully accounted for in the PTM, and therefore, this variable was placed as the first indicator of the model.
Age has been reported as a predictor of brain injury outcome in many studies, including the present study.5–8,10,15,22 However, the relationship between age and mortality rate has been reported as nonlinear, and the mortality rate is markedly higher in octogenarians and nonagenarians than in any other age groups.6 On the other hand, the relationship between age and functional recovery found in the present study was inverse, that is, older people were more prone to show poor functional recovery than were younger people.
In addition, our results show that subarachnoid hemorrhage and systolic blood pressure at admission are significant predictors of functional recovery at the acute and postacute stages, respectively. The presence of subarachnoid hemorrhage adversely affected brain injury outcomes in other studies,29 and blood pressure is also associated with brain injury outcome, that is, a lower blood pressure at admission is a predictor of a poorer recovery.29–31 We found that systolic blood pressure is a significant predictor of 6-month functional disability; however, this relationship was nonlinear. Specifically, 6-month functional disability of patients with a systolic blood pressure of less than 150 mm Hg on admission improved with increases in systolic blood pressure, but the 6-month functional disability of those with a systolic blood pressure of 150 mm Hg or more at admission became worse with increases in systolic blood pressure.
In the present study, patients with an admission systolic blood pressure in the range of 120 to 150 mm Hg showed the best 6-month functional recoveries. This result seems to be meaningful in terms of proposing preferable ranges of systolic blood pressure for a good prognosis. The fact that initial systolic blood pressure is significantly associated with long-term recovery is clinically meaningful, because an abnormal blood pressure can be accurately and promptly detected and corrected.
In the present study, pupillary reflex and heart rate were identified as significant predictors of 1-month cognitive recovery, whereas pupillary reflex, heart rate, and midline shift were identified as significant predictors of 6-month cognitive recovery. That is, good cognitive recovery appears to be commonly associated with a better pupillary reflex and a lower heart rate in the acute and post-acute stages of TBI. Pupillary reflex has been repeatedly reported to be the most important predictor of outcome after brain injury.7,8,29,32 Less frequently, heart rate and midline shift have been identified as significant predictors.6,8
Study Limitations
The general applicability of our results is limited by the small sample size and the uneven distribution of PTM scores. Therefore, further studies of subjects with a wide range of PTM scores in large samples are needed to confirm our findings. Additional studies are also needed to determine whether significant predictors differ according to the severity of the TBI, which was not examined in our study. In addition, a potential for bias exists because all instruments were administered by only 1 of the investigators.
Nursing Implications
For critical care nurses, the most important aims of the nursing care for patients with TBI are to decrease mortality and improve functional and cognitive outcomes. Accurate nursing assessment is key to achieving this goal. Simple and accurate predictive models with easily assessable variables are required. Thus, developing and verifying predictive models for TBI are important areas of nursing research. The present study is likely to be meaningful in terms of model verification and accordingly provides the basic frame of evidence-based nursing care for patients with TBI in the acute period. In addition, the findings from this study may contribute to the evidence on the importance of acute assessment and management of GCS score, pupillary reflex, systolic blood pressure, and heart rate for short- and long-term functional and cognitive recovery after TBI.
Conclusions
PRIME POINTS
- A prediction model for recovery or mortality after brain injury must be quick and simple enough to use without special training.
- The degree of recovery in patients with traumatic brain injury depends on acute medical management as well as many other factors.
- Is the Prediction Tree Model better than the general model for predicting functional and cognitive recovery at 1 month or 6 months?
Footnotes
- To purchase electronic or print reprints, contact The InnoVision Group, 101 Columbia, Aliso Viejo, CA 92656. Phone, (800) 899-1712 or (949) 362-2050 (ext 532); fax, (949) 362-2049; e-mail, [email protected].
- Financial Disclosures
This work was supported by a research grant from Inha University, Inchon, Republic of Korea. The Inchon Landing during the Korean War broke the strangle hold the North Koreans had on the Allied forces in the Pusan Perimeter sadly because of an overreach the results of this brilliant maneuver failed to yield the results